
Friday the FDA (Food and Drug Administration) sent out a warning about high doses of ondansetron (Zofran). The doses are not the kind of doses used in EMS, but have been used to treat nausea and vomiting from chemotherapy.
GlaxoSmithKline (GSK) has announced changes to the Zofran drug label to remove the 32 mg single intravenous dose. The updated label will state that ondansetron can continue to be used in adults and children with chemotherapy-induced nausea and vomiting at the lower intravenous dose recommended in the drug label, a dose of 0.15 mg/kg administered every 4 hours for three doses; however, no single intravenous dose should exceed 16 mg. Information from the new clinical study will be included in the updated drug label.[1]
The usual EMS/ED dose is 4 mg, so this does not directly affect the way we use ondansetron. However, we may see patients treated with much larger doses, so we should be aware of the potential complications.
Here is the not yet changed adult chemotherapy dosing information from the label –
2.1 Prevention of Nausea and Vomiting Associated with Initial and Repeat Courses of Emetogenic Chemotherapy
ZOFRAN Injection should be diluted in 50 mL of 5% Dextrose Injection or 0.9% Sodium Chloride Injection before administration.
Adults
The recommended adult intravenous dosage of ZOFRAN is a single 32-mg dose or three 0.15-mg/kg doses. A single 32-mg dose is infused over 15 minutes beginning 30 minutes before the start of emetogenic chemotherapy. Efficacy of the 32-mg single dose beyond 24 hours has not been established. The recommended infusion rate should not be exceeded [see Overdosage(10)]. With the three-dose (0.15-mg/kg) regimen, the first dose is infused over 15 minutes beginning 30 minutes before the start of emetogenic chemotherapy. Subsequent doses (0.15 mg/kg) are administered 4 and 8 hours after the first dose of ZOFRAN.[2]
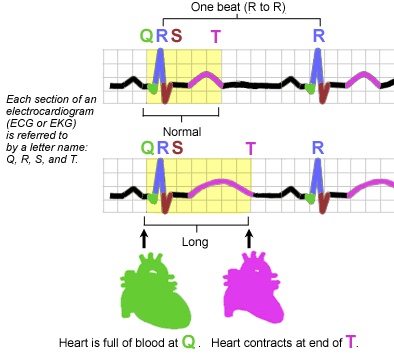
What does QT segment prolongation look like?
There is a problem with the image. The ventricles contract during the QRS complex, not during the T wave.
- The use of a single 32 mg intravenous dose of ondansetron should be avoided. New information indicates that QT prolongation occurs in a dose-dependent manner, and specifically at a single intravenous dose of 32 mg.
- Patients who may be at particular risk for QT prolongation with ondansentron are those with congenital long QT syndrome, congestive heart failure, bradyarrhythmias, or patients taking concomitant medications that prolong the QT interval
- Electrolyte abnormalities (e.g., hypokalemia or hypomagnesemia) should be corrected prior to the infusion of ondansetron.
- The lower dose intravenous regimen of 0.15 mg/kg every 4 hours for three doses may be used in adults with chemotherapy-induced nausea and vomiting. However, no single intravenous dose of ondansetron should exceed 16 mg due to the risk of QT prolongation.
- The new information does not change any of the recommended oral dosing regimens for ondansetron, including the single oral dose of 24 mg for chemotherapy induced nausea and vomiting.[1]
There are many drugs that prolong the QT segment. There are three lists of these drugs broken down by potential to cause torsades, from qtdrugs.org.[4]
Some of these drugs are given for VT (Ventricular Tachycardia) because of a lack of awareness of the QT segment prolonging effects of antiarrhythmics. When will amiodarone receive some sort of warning for the same problem, especially since amiodarone is given like candy to patients with cardiac conduction disorders?
I will look at the problems with amiodarone and the ACLS recommendation that amiodarone be given to patients with polymorphic VT (Ventricular Tachycardia) in Part II.
–
The study referenced by the FDA does not appear to be available, yet. It is listed as completed –
ClinicalTrials.gov Identifier: NCT01449188
–
Footnotes:
–
[1] Ondansetron (Zofran) IV: Drug Safety Communication – QT prolongation
FDA
FDA Safety Information and Adverse Event Reporting Program Safety Information
[Posted 06/29/2012]
Safety Alert
–
[2] ZOFRAN (ondansetron hydrochloride) injection
[GlaxoSmithKline LLC]
FDA label
Label
–
[3] QT Drug Lists by Risk Groups
qtdrugs.org
Home page
.


[…] [4] Ondansetron (Zofran) Warning for QT Prolongation – is Amiodarone next? – Part I Mon, 02 Jul 2012 Rogue Medic Article […]