I don’t expect to see this as a headline anywhere, but this possible cause of death something we should be aware of.
Abdominal pain in a patient with many comorbidities. She is given medication and later is found dead at her home.
What drugs was she taking?
a Potentially proarrhythmic drugs as classified by the Arizona Center for Education and Research on Therapeutics (www.qtdrugs.org).
b Given during emergency department visit.[1]
What risk factors did she have?
a Risk factors present in case study.[1]
The 12 lead obtained in the ED (Emergency Department) shows a bradycardia with a heart rate of 58 beats per minutes. Bradycardia probably should have been included in the risk factors in this case.
What treatment did she receive that increased her risk of TdP (torsades de pointes)?
Ondansetron (Zofran)
On September 15, 2011, the FDA issued a Medwatch Safety Alert for Zofran (ondansetron) in patients with congenital Long QT syndrome, a heart arrhythmia. The FDA further required GlaxoSmithKline to conduct a thorough QT study to determine the degree to which Zofran may cause QT interval prolongation.[1] On June 29, 2012, the FDA issued an FDA Drug Safety Communication Update entitled New information regarding QT prolongation with ondansetron (Zofran).
The 32-mg high dose of ondansetron (Zofran) has been pulled from the market by the FDA because of concerns about cardiac problems.[15][2]
The high dose of 32 mg is more than would usually be given by EMS or in the ED.
In this case, the ondansetron was 8 mg given orally in the ED, so this was a much smaller dose.
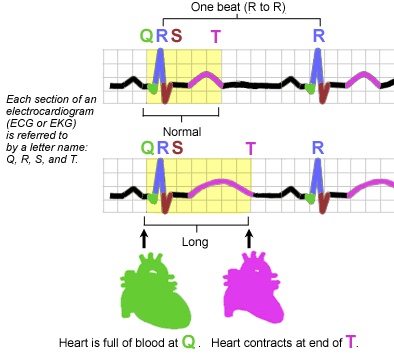
What is QT segment prolongation?
There is a problem with the image. The ventricles contract during the QRS complex, not during the T wave.
Let’s see some torsades.

Click on images to make them larger.[3]
How do we know that it is TdP?
Because of the long QT segment in the beats preceding the VT (Ventricular Tachycardia).
Does all torsades go away on its own, as the above example did?
No.
A medical screening examination was conducted and 8 mg of orally disintegrating ondansetron (Zofran) was administered for persistent nausea and vomiting. A 12-lead electrocardiogram (ECG) completed at triage (Figure 1) was remarkable for left ventricular hypertrophy and QT interval prolongation.[1]
This is a patient who should be on a monitor, not necessarily because of the proarrhythmic effects of the drugs she is already taking, but because of the combination with the proarrhythmic drug she has been given in the ED.
Shortly thereafter, the patient self-discharged from the emergency department before receiving definitive treatment. Upon making a follow-up phone call, it was discovered that the patient had been found unresponsive in bed approximately 4 hours after leaving the emergency depart[1]
Was it the Zofran?
Maybe, but if it was, the ondansetron may only be the straw that broke the camel’s back.
Does that mean that the risks should have been ignored?
No.
Many of the patients we see are the most fragile people in society and we are seeing them when they are at their greatest vulnerability to adverse treatment effects.
The FDA has warned about the QT prolonging effects of ondansetron, so we cannot claim that we could not have known. I have written about this before.[4],[5]
We should be looking for reasons why we should not be giving treatments.
EMS operates under protocols that may state –
If condition X is present, give treatment A, then give treatment B.
ED treatment can be just as protocol driven as EMS treatment.
We have drugs that can be dangerous under certain circumstances.
Should we give any drug without considering the possible drug interactions and adverse events?
If we are not aware of the drug’s possible drug interactions and adverse events, should we be permitted to give the drug?
Ondansetron is one of the drugs I give frequently, but I need to remind myself to consider the possible QT prolonging effects and to look for other QT prolonging drugs and medical conditions.
What other drugs do I carry that can cause QT prolongation?
Amiodarone (Nexterone, Cordarone) is the only drug I carry that is on the Drugs with a Risk of Torsades de Pointes list. Even ondansetron is not on this list. There is less evidence that ondansetron causes torsades, than there is that amiodarone causes torsades.
Substantial evidence supports the conclusion that these drugs prolong the QT interval and have a risk of TdP when used as directed in labeling.[6]
Oxytocin (Pitocin) and ondansetron are on the Drugs with a Possible Risk of Torsades de Pointes list.
Substantial evidence supports the conclusion that these drugs cause QT prolongation but there is insufficient evidence that they, when used as directed in labeling, have a risk of causing TdP.[6]
Diphenhydramine (Benadryl) is the only drug I carry that is on the Drugs with a Conditional Risk of Torsades de Pointes list.
Substantial evidence supports the conclusion that these drugs prolong the QT interval and have a risk of TdP but only under certain known conditions (e.g. excessive dose, drug interaction, etc.).[6]
All of these drugs are generally considered to be safe, because we are ignorant of the adverse events they can cause. TdP is only one adverse event. Amiodarone has other proarrhythmic effects, can cause hypotension,
Then there are the many drugs that may interact with these drugs to prolong the QT segment.
Antibiotics, psychiatric medications (for all kinds of psychiatric conditions – psychosis to depression), erectile dysfunction drugs,
Then why aren’t we seeing large numbers of dead bodies?
These patients have other medical conditions that may lead to death without any TdP or there may not be much TdP caused by these drugs. We do not know.
We do know that thousands, even tens of thousands of patients can die without anyone noticing that the deaths are the effect of a drug.[7]
For example –
She was pronounced dead at the scene by emergency care providers. Because of her extensive medical history, the woman’s family declined an autopsy, and her primary physician attributed the death to complications of diabetes mellitus, end-stage renal disease, and hypertension.[1]
What was the cause of death?
We do not know.
Given the number of risk factors for TdP, is TdP a likely cause?
Torsades de pointes is no less likely a cause of death than anything listed by the her primary care physician on the death certificate.
This isn’t a multiple choice exam, where someone thinks that there is some mythological one best answer.
This is the real world and all of these conditions probably significantly contributed to her death.
Was ondansetron the final straw?
Maybe.
–
Footnotes:
–
[1] Woman with Risks for Torsades de Pointes Dying within Hours of Leaving the Emergency Department.
Pickham D, Sickler K.
J Emerg Nurs. 2011 Dec 2. [Epub ahead of print] No abstract available.
PMID: 22137882 [PubMed – as supplied by publisher]
–
[2] Ondansetron
Wikipedia
Adverse effects
Article
–
[3] Etiology, warning signs and therapy of torsade de pointes. A study of 10 patients.
Keren A, Tzivoni D, Gavish D, Levi J, Gottlieb S, Benhorin J, Stern S.
Circulation. 1981 Dec;64(6):1167-74.
PMID: 7296791 [PubMed – indexed for MEDLINE]
Abstract with link to Free Full Text Download in PDF format from Circulation
–
[4] Ondansetron (Zofran) Warning for QT Prolongation – is Amiodarone next? – Part I
Mon, 02 Jul 2012
Rogue Medic
Article
–
[5] Ondansetron (Zofran) Warning for QT Prolongation – is Amiodarone next? – Part II
Thu, 05 Jul 2012
Rogue Medic
Article
–
[6] TdP drug lists
AZCERT.org is now CredibleMedsTM
Web page with links to all lists as pop ups.
–
[7] Mortality and morbidity in patients receiving encainide, flecainide, or placebo. The Cardiac Arrhythmia Suppression Trial.
Echt DS, Liebson PR, Mitchell LB, Peters RW, Obias-Manno D, Barker AH, Arensberg D, Baker A, Friedman L, Greene HL, et al.
N Engl J Med. 1991 Mar 21;324(12):781-8.
PMID: 1900101 [PubMed – indexed for MEDLINE]
Free Full Text Article from N Engl J Med.
.


Someone dying after going home AMA from the ED wasn’t an interesting feature of the article. Nor was the possible causative factor of a prolonged QT.
What was interesting was this tidbit: “After the drug was administered and electrocardiography was performed, the patient was returned to the waiting room to await further treatment.”
I wonder how much of a role that plays in a patient’s decision to leave AMA.
Seems bizarre that a patient with as many comorbid factors who shows a long QT and hypokalemia (I assume they drew labs, they were not listed in the paper) who goes to dialysis 3 times a week was allowed to go back to the waiting room given her initial exam.
Christopher,
I have to cut some stuff out, otherwise everything will be 4,000 words long. I figured that sentence alone could result in a thousand words.
–
California is known for its waiting times, so she may have been there a long time before she was seen. She might have felt better. She might have felt that the message was Zofran is all she would get for her abdominal pain. She might have remembered that she had something cooking in the oven. We do not know.
–
Even without all of the risk factors, a current QTc of 538 is not proof of anything, but probably much more deserving of a monitored bed than many of the presentations of patients who are on a monitor for whatever.
.
–
Maybe there was some animosity somewhere. Animosity encourages us to make stupid decisions.
When we base our patient care on disliking a patient (or someone else), we provide bad patient care.
When we base our care on liking a patient, we provide bad patient care.
Maybe they were packed. Maybe the nurse did not like the patient. Maybe that is hospital policy. Maybe . . . .
I am pleased that they decided to write about this. Voluntarily reporting potentially preventable bad outcomes is not as common as it should be.
.
I agree with Christopher here. That’s does seem a bit of a bizarre that she was admitted back to the waiting room after her initial exam.
I think the evidence is a little thin to start implicating the ondansetron just yet. In addition, a few elements of the case report are odd. In no particular order:
– They list hypo-K as “present in case study,” but I don’t find any indication that labs were drawn. It’s entirely possible that she had hypEr-K, given her dialysis schedule.
– We don’t have an old ECG. Was the QTc change new or old? Or even improved from prior?
– They mention a “medical screening exam,” which in EMTALA language usually means a physician/midlevel exam. The context suggests that only the triage nurse saw the patient.
A case study without labs or an autopsy is unfulfilling, and sort of pointless, What about aortic aneurysm? NSTEMI? Mesenteric ischemia? This is all philosophizing.
As for sending the patient out to the waiting room, it happens. I personally don’t like the idea of putting an ESRD/DM patient with abdominal pain back in the waiting room, but the ECG was negative, and she may have looked really good. Or she looked terrible, and triage blew her off due to counter-transference (psych patients have have it tough in our society).
As for for ondansetron, let’s not throw the baby out with the droperidol!
Brooks,
Yes, the evidence is thin, but I wrote about this as something to be aware of, which many people may not even consider.
–
2 days after dialysis, hypokalemia is improbable, but we do not know if labs were drawn. As with writing blog posts, changing details to protect the patient’s privacy may be part of the story.
–
With her history, it is likely that she has had several previous 12 lead ECGs. This would be useful information.
–
Parts of the story appear contradictory. This, the hypokalemia, and the bradycardic 12 lead with no mention of bradycardia. The patient has two lower limb amputations, but self discharged. With her history, I am surprised that she would have been living outside of a nursing home, but living independently is suggested by the story.
The drug list seems like a typical nursing home drug list, except for the absence of laxatives and pain medicine.
–
This is true, but that is not the point of what I wrote.
GlaxoSmithKline did a post approval study of QT prolongation that led to the removal of the 32 mg dose from approved treatments.
http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm310219.htm?source=govdelivery
Healthy subjects – how relevant is that to our patient?
Maybe not even a little bit.
The Clinical Trials registration points out some of the problems with understanding the possible adverse events.
http://clinicaltrials.gov/ct2/show/NCT01449188
The FDA approved label does mention dose-dependent QT prolongation as caused by ondansetron and TdP as associated with ondansetron.
http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=d9a71b42-ddfc-49d5-7280-0fc0041dba41#section-7.2
This is a patient with a longer than normal QT segment. She has several risk factors for QT segment prolongation. She was given a drug that is known to prolong the QT segment. Even the label for Zofran ODT recommends ECG monitoring of patient with even one of several risk factors – she was reported to have most of the risk factors.
http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=c7d61d98-fe86-4340-9b86-47eb92acaa0e
–
Is a QTc of 538 really negative?
–
I started writing a post on this topic in my response to Christopher. It is an important topic, but where to stop ranting.
–
I never suggested that we should eliminate, or even further limit the recommendations for ondansetron.
I am strongly recommending better familiarity with the information that is on the label and a greater awareness of the potential adverse events that might occur.
I think that this is dramatically different from the continuous 12 leads prior to administration through to hours after elimination that is recommended by the black box warning for droperidol, even when used for excited delirium.
Ondansetron is a safe drug, when given appropriately. the same is true of oxygen. 😉
.
I don’t know Brooke, seems like a malpractice suit waiting to happen…but it probably happens more than it should…scary.
Christopher is right.. That’s a little bit strange.